Front. Pharmacol., Sec. Ethnopharmacology, 2022
Background:
Morus alba L. (Sangzhi) alkaloids (SZ-A), extracted from the Chinese herb Morus alba L. (mulberry twig), have been shown to ameliorate hyperglycemia in type 2 diabetes and have been approved for diabetes treatment in the clinic. However, their versatile pharmacologic effects and regulatory mechanisms are not yet completely understood.
Purpose:
This study explored the protective effects of SZ-A on islet β cells and the underlying mechanism.
Methods:
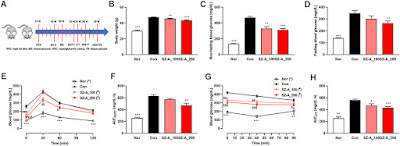
Type 2 diabetic KKAy mice were orally administered SZ-A (100 or 200 mg/kg, once daily) for 11 weeks, and oral glucose tolerance, insulin tolerance, intraperitoneal glucose tolerance and hyperglycemia clamp tests were carried out to evaluate the potency of SZ-A in vivo. The morphology and β-cell dedifferentiation marker of KKAy mouse islets were detected via immunofluorescence. The effect of SZ-A on glucose-stimulated insulin secretion was investigated in both the islet β-cell line MIN6 and mouse primary islets. Potential regulatory signals and pathways in insulin secretion were explored, and cell proliferation assays and apoptosis TUNEL staining were performed on SZ-A-treated MIN6 cells.
Results:
SZ-A alleviated hyperglycemia and glucose intolerance in type 2 diabetic KKAy mice and improved the function and morphology of diabetic islets. In both MIN6 cells and primary islets, SZ-A promoted insulin secretion. At a normal glucose level, SZ-A decreased AMPKα phosphorylation, and at high glucose, SZ-A augmented the cytosolic calcium concentration. Additionally, SZ-A downregulated the β-cell dedifferentiation marker ALDH1A3 and upregulated β-cell identifying genes, such as Ins1, Ins2, Nkx2.2 and Pax4 in KKAy mice islets. At the same time, SZ-A attenuated glucolipotoxicity-induced apoptosis in MIN6 cells, and inhibited Erk1/2 phosphorylation and caspase 3 activity. The major active fractions of SZ-A, namely DNJ, FAG and DAB, participated in the above regulatory effects.
Conclusion:
Our findings suggest that SZ-A promotes insulin secretion in islet β cells and ameliorates β-cell dysfunction and mass reduction under diabetic conditions both in vivo and in vitro, providing additional supportive evidence for the clinical application of SZ-A.